Zirconia ceramics are notoriously prone to micro-cracking and warping during the cooling phase, issues often undetected until final inspection. A single fracture renders an entire batch useless, wasting expensive high-purity powder and derailing tight production schedules. By strictly controlling thermal gradients, binder burnout, and pressure distribution, you can achieve defect-free density every time. With years of material science expertise, we utilize these exact protocols to ensure component integrity for our clients.
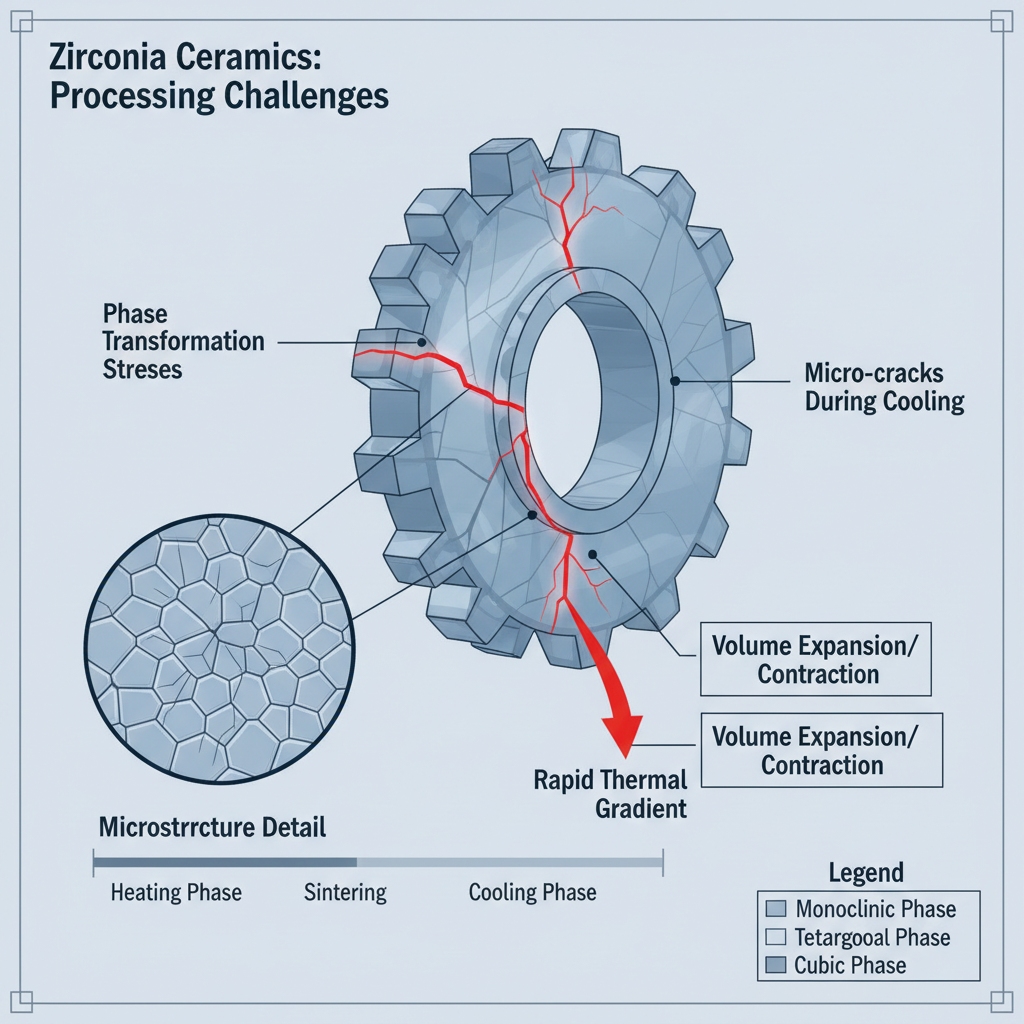
What Challenges Arise in Zirconia Ceramics?
Phase transformation drives internal stress that compromises structural integrity.
What drives phase transformation?
Zirconia shifts from a tetragonal to a monoclinic phase as it cools, a process highly sensitive to thermal conditions. This transformation involves significant volume expansion.
- Volume expansion risks
- Internal stress generation
- Structural integrity loss
How do thermal fluctuations impact integrity?
Here is the main issue. Rapid temperature changes induce immediate stress within the ceramic matrix. This leads to irreversible structural damage before the part even leaves the furnace.
Key Takeaway
Managing phase changes protects the component’s final integrity. By controlling the thermal environment, you prevent the internal stresses that tear the material apart from the inside out.
| Factor | Impact on Quality | |
|---|---|---|
| Phase Change | High risk of internal stress | |
| Thermal Control | Prevents irreversible damage | |
| Stability | Ensures functional integrity |
Precise thermal management is the only defense against phase-related defects.
How Critical is Powder Selection for Zirconia Ceramics?
Purity directly dictates the mechanical strength and sintering behavior of the final part.
Why does purity matter?
Impurities like oxides or metals act as stress concentration points within the matrix. These contaminants weaken the ceramic and create uneven densification.
- Reduces mechanical strength
- Creates uneven densification
- Introduces vulnerabilities
Does particle size distribution affect density?
But wait, there’s more. Uniform shrinkage relies heavily on a narrow particle size distribution. An imbalance of fines and coarse particles leads to localized density variations.
Key Takeaway
High purity minimizes stress points in the finished product. Selecting the right powder ensures that shrinkage occurs uniformly, preventing the internal tension that causes cracks.
| Powder Trait | Effect on Sintering | |
|---|---|---|
| High Purity | Minimizes stress points | |
| Narrow PSD | Promotes uniform shrinkage | |
| Balanced Fines | Prevents localized low density |
Invest in high-grade powder to eliminate defects at the source.

How Should You Handle Zirconia Ceramics Powder?
Moisture and contaminants absorbed during storage ruin sintering performance.
Can storage conditions cause defects?
Improper storage exposes powder to environmental humidity and debris. Even microscopic contaminants entered during batching can result in catastrophic failure.
How does moisture impact sintering?
You need to know this. Moisture compromises the purity of the binder system and the powder itself. This leads to steam generation during heating, which cracks the green body.
- Keep containers sealed
- Control humidity levels
- Avoid cross-contamination
Key Takeaway
Proper storage blocks moisture absorption effectively. Maintaining a dry, controlled environment preserves the flowability and chemical integrity of your raw materials.
| Action | Prevention Benefit | |
|---|---|---|
| Sealed Storage | Blocks moisture absorption | |
| Clean Batching | Stops metallic contamination | |
| Humidity Control | Maintains powder flowability |
Strict handling protocols are non-negotiable for defect-free ceramics.
What Ensures Uniformity in Green Body Formation?
Uniform pressure distribution prevents density gradients that lead to warping.
Why is uniform pressure essential?
Uneven pressure during molding creates areas of varying density within the green body. These gradients cause differential shrinkage during sintering, pulling the part out of shape.
How does CIP prevent warping?
Here is the trick. Cold Isostatic Pressing (CIP) applies pressure from all directions simultaneously. This eliminates the density variations common in standard die pressing.
- Equal pressure distribution
- Consistent green density
- Reduced structural imbalances
Key Takeaway
CIP ensures consistent shrinkage across the entire geometry. By standardizing density, you eliminate the primary mechanical cause of warping during the heating phase.
| Method | Resulting Quality | |
|---|---|---|
| Uniform Pressure | Consistent shrinkage | |
| CIP Technology | Reduced density gradients | |
| Mold Calibration | Prevents geometric distortion |
Adopt isostatic pressing to guarantee geometric stability.

Why is Binder Removal Critical for Zirconia Ceramics?
Slow removal prevents gas bloating and the formation of internal voids.
What happens if debinding is too fast?
Rapid heating causes the binder to vaporize faster than it can escape. This trapped gas creates pressure pockets that bloat or crack the fragile green body.
How do you prevent internal voids?
Think about this. A gentle ramp rate of 1-2°C/min allows gases to diffuse harmlessly. The critical range is typically between 200°C and 400°C.
- Monitor exhaust gas
- Extend hold times
- Allow gas escape paths
Key Takeaway
A slow ramp stops cracking caused by gas pressure. Patience during the debinding phase is critical to preserving the internal structure of the ceramic.
| Process | Risk Mitigation | |
|---|---|---|
| Slow Ramp | Prevents bloating/cracking | |
| Extended Hold | Ensures complete burnout | |
| Optimized System | Eliminates residual carbon |
Optimize your debinding cycle to protect the green body.
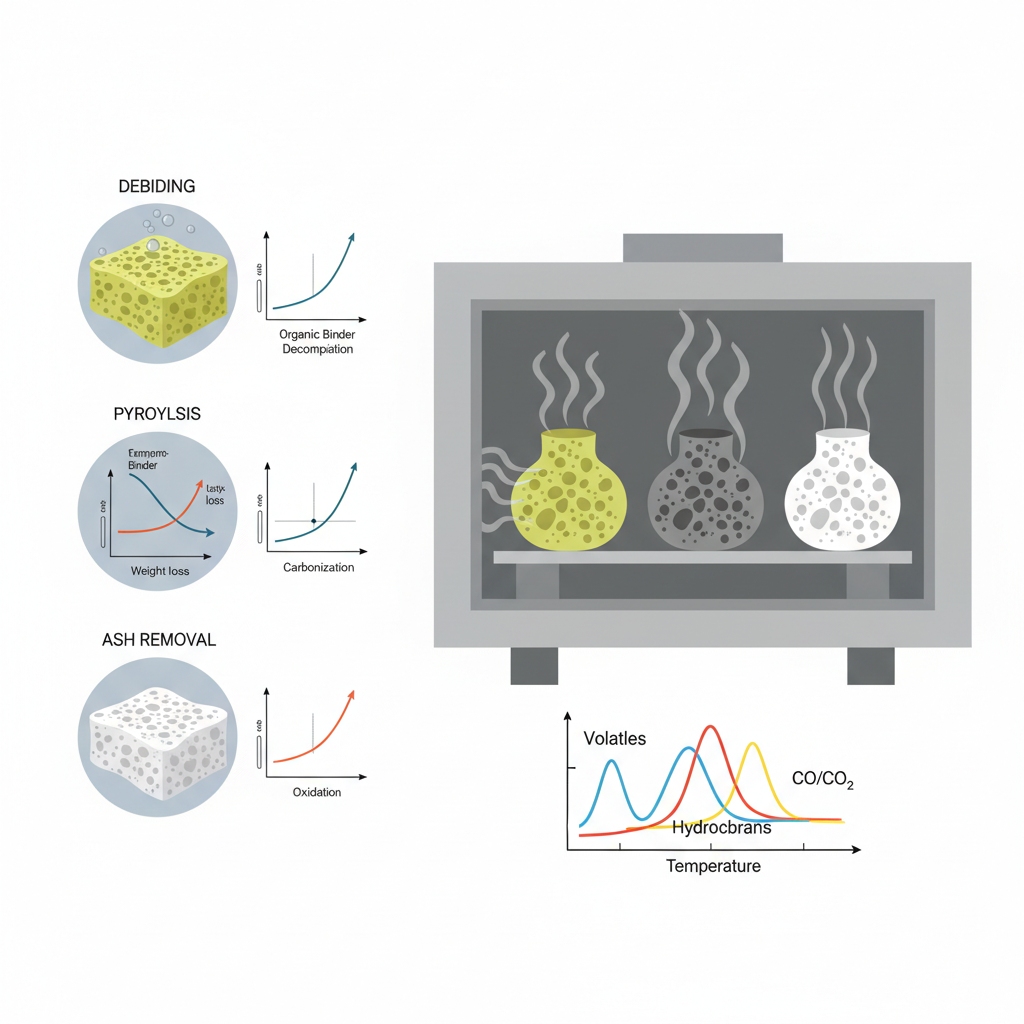
How Do You Optimize the Heating Ramp?
Gradual heating avoids thermal shock while ensuring complete particle bonding.
What is the ideal heating rate?
Heating rates must be controlled to prevent thermal shock as the material densifies. A smooth transition into sintering temperatures allows grains to arrange without stress.
Why is the soak time important?
Don’t miss this. The soak time balances particle bonding against unwanted grain growth. Too long, and grains coarsen; too short, and porosity remains.
- Too short: Residual porosity
- Too long: Grain coarsening
- Optimal: Maximum density
Key Takeaway
An optimized soak achieves full density without degrading material properties. Precise time management at peak temperature is the key to a strong microstructure.
| Phase | Outcome Control | |
|---|---|---|
| Gradual Ramp | Avoids thermal shock | |
| Peak Soak | Achieves full density | |
| Time Management | Preserves grain structure |
Tune your sintering curve for maximum density.

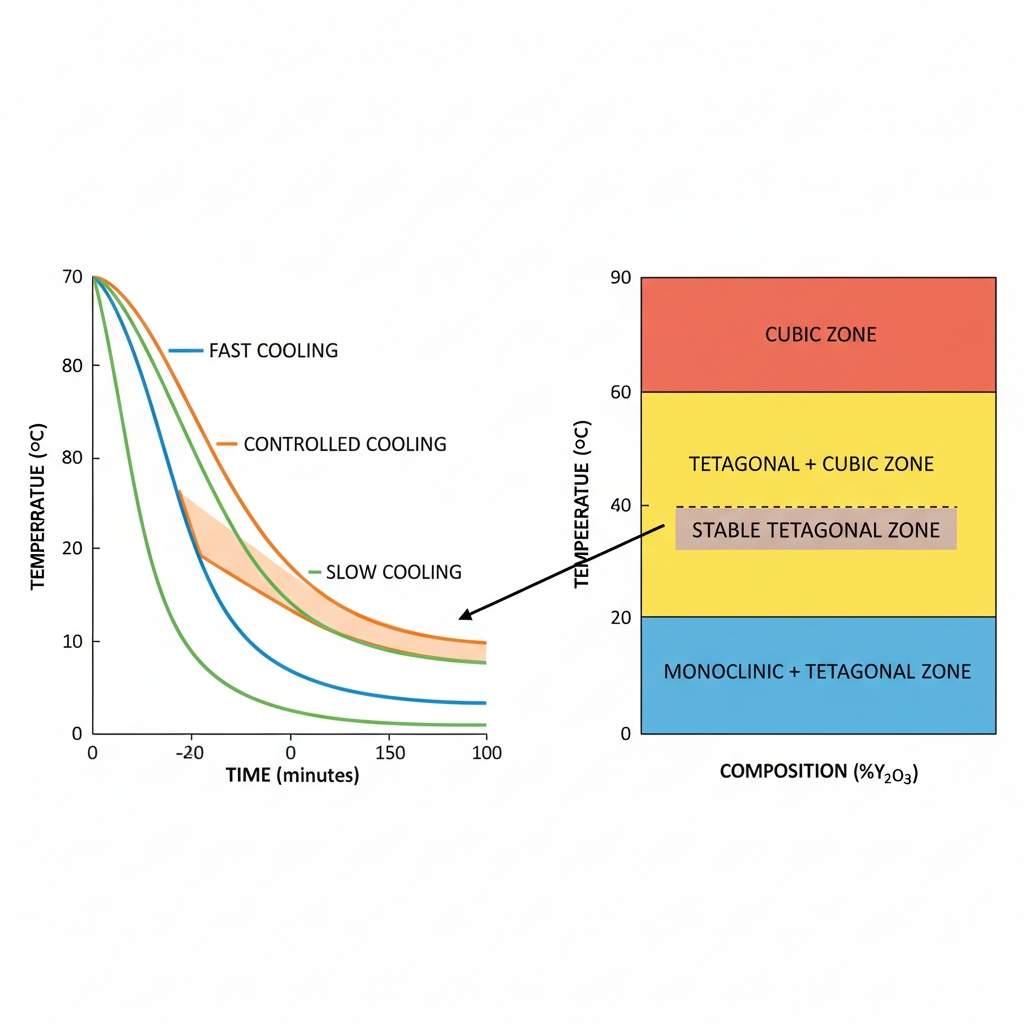
Why is the Cooling Phase Risky for Zirconia Ceramics?
The transition at 1170°C triggers a phase change that generates immense internal stress.
What occurs at the 1170°C transition?
This is the critical point where the material attempts to transform from tetragonal to monoclinic. This shift involves volume changes that can shatter the ceramic if not managed.
Can rapid cooling cause fractures?
Be warned. Rapid cooling through this zone guarantees catastrophic failure. The outer surface cools faster than the core, creating tension that snaps the part.
- Slow rate at 1170°C
- Controlled transition
- Monitoring internal temp
Key Takeaway
Slow cooling prevents fractures by equalizing temperature gradients. You must maintain a controlled descent through the transformation zone to ensure survival.
| Temp Range | Action Required | |
|---|---|---|
| >1170°C | Moderate cooling allowed | |
| ~1170°C | Slow, controlled cooling | |
| <1000°C | Stabilize to room temp |
Never rush the cooling process at critical thresholds.
Does Furnace Atmosphere Affect Zirconia Ceramics?
Atmosphere stability prevents surface reactions that degrade aesthetics and strength.
How does oxidation impact performance?
An uncontrolled oxidizing environment can lead to unwanted chemical reactions on the surface. This often results in discoloration or a weakened outer layer.
Is a vacuum environment necessary?
Here is the deal. While some ceramics require vacuums, zirconia typically needs a stable, clean air environment. Contaminants in the furnace atmosphere must be rigorously excluded.
- Atmosphere stability
- Contamination prevention
- Material compatibility
Key Takeaway
Controlled air is standard for most zirconia applications. Keeping the furnace atmosphere clean ensures the material retains its intended chemical composition.
| Condition | Material Impact | |
|---|---|---|
| Oxidizing | Potential surface reactions | |
| Controlled Air | Standard for many oxides | |
| Vacuum | Specialized application use |
Maintain a clean furnace atmosphere to protect surface quality.
What Are Common Zirconia Ceramics Defects?
Cracks and warping primarily stem from thermal shock and uneven support.
What causes surface cracks?
Surface cracks are usually the result of rapid cooling or residual stress from molding. They appear when the tension on the surface exceeds the material’s strength.
Why does warping occur?
You might ask… Warping happens when the part is not supported evenly. Friction against the setter plate during shrinkage can also drag and distort the shape.
- Use flat setter plates
- Check furnace heating balance
- Verify green body density
Key Takeaway
Proper support prevents warping during the shrinkage phase. Using high-quality alumina setter plates reduces friction and keeps the geometry true.
| Defect | Primary Cause | |
|---|---|---|
| Surface Cracks | Fast cooling / Shock | |
| Warping | Uneven density / Support | |
| Bloating | Incomplete debinding |
Address support and cooling to eliminate common physical defects.

How Can You Ensure Holistic Quality Control?
A systemic approach tracks every variable from raw powder to final cooling.
Why is a systemic approach necessary?
Production is an interconnected system where a small error upstream amplifies downstream. You cannot fix a molding error in the sintering furnace.
How do you monitor environmental conditions?
The bottom line. You must track temperature, humidity, and pressure at every stage. Consistent data logging allows you to pinpoint the exact moment a defect is introduced.
- Raw material inspection
- Green body auditing
- Furnace calibration
Key Takeaway
Monitoring guarantees quality by catching deviations early. A rigorous QC protocol is the only way to ensure repeatable, profitable manufacturing.
| Stage | Control Measure | |
|---|---|---|
| Material | Purity Certification | |
| Forming | Pressure Calibration | |
| Sintering | Curve Optimization |
Implement total process control for defect-free results.

Conclusion
Preventing cracks requires a balance of high-purity powder, uniform green body formation, and a rigorously controlled sintering curve—especially during cooling. By mastering these variables, you ensure repeatable quality and profitability. Empowering precision manufacturing for a defect-free future is our mission. Struggling with warping? Contact Global Industry today for a complimentary consultation or to request a sample of our high-stability zirconia powder.
FAQ Section
1. Can rapid cooling immediately ruin zirconia ceramics?Yes, specifically around 1170°C. Rapid cooling through this zone triggers a sudden phase transformation that creates massive internal stress, leading to immediate fracturing or catastrophic failure of the component.
2. How does binder choice affect cracking in zirconia ceramics?If the binder system doesn’t match the powder or burn out cleanly, it creates gas pockets. These pockets result in bloating and internal voids that severely weaken the structure and cause cracks during sintering.
3. What is the best way to support zirconia ceramics during sintering?Use high-quality, flat alumina setter plates. This provides uniform support across the base of the part and prevents the component from sagging or warping due to friction or uneven heating during shrinkage.
4. Can I fix a warped green body before sintering?Generally, no. If a green body has significant density gradients from the pressing stage, it will warp during sintering regardless of furnace controls; you must optimize the pressing stage to fix the root cause.
5. Why do I see surface cracks even with slow cooling?This often points to issues upstream. It typically indicates residual stress from the molding process or impurities in the raw powder that act as stress concentrators, which manifest as cracks even under ideal cooling conditions.