Unpredictable raw material variations frequently sabotage ceramic glaze finishes, leaving production managers scrambling to fix ruined batches. These sudden color mismatches trigger expensive downtime, force reformulation, and threaten your brand’s reputation for premium quality. This guide analyzes the technical drivers of Zirconium Silicate color instability and details the manufacturing protocols necessary to secure a consistently white, high-purity supply. For a related discussion on quality, see our article on How Can You Achieve Better Glaze Smoothness And Gloss With Zirconium Silicate Powder?
Assessing Zirconium Silicate Quality Standards

Does aesthetic consistency determine value?
Visual uniformity is the primary non-destructive indicator of chemical purity and directly governs the market pricing of your final ceramic products. In sectors like sanitary ware, even microscopic deviations in whiteness can lead to immediate batch rejection during quality control. Here is the deal: if your raw powder fluctuates, your end product loses its competitive edge.
- Market pricing power
- Consumer brand perception
- Glaze application uniformity
Why does color failure occur?
Color failure usually stems from overlooked trace contamination, resulting in production bottlenecks as teams rush to reformulate recipes on the fly. You might be wondering how these seemingly minor visual defects translate into tangible financial losses for your operation.
What is the cost of inconsistency?
Inconsistent raw materials force operational teams to waste valuable hours adjusting kiln curves and glaze formulas rather than focusing on output. The bottom line is that stability is an economic necessity, not just a visual preference.
| Defect Type | Operational Consequence | Financial Risk Profile | |
|---|---|---|---|
| Grey Undertone | Complete glaze reformulation | High (Significant Downtime) | |
| Black Spotting | Immediate batch disposal | Severe (Scrap & Waste Costs) | |
| Yellowing | Product class downgrading | Moderate (Margin Erosion) |
Key Takeaway: Consistent color stability is the foundational metric for operational efficiency and profitability.
Identifying Impurities in Zirconium Silicate

How does iron oxide degrade whiteness?
Iron oxide (Fe2O3) acts as the most aggressive contaminant, introducing persistent yellow, pink, or reddish-brown chromophores even at parts-per-million levels. But here is the catch: these metallic impurities often remain dormant in raw powder and only fully manifest their damaging colors during the high-temperature oxidation of the kiln firing process.
- Fe2O3 (Reddish-Brown)
- Fe3O4 (Black Magnetic)
- Free Metallic Iron (Grey)
Does titanium amplify discoloration?
Titanium Dioxide (TiO2) frequently co-exists with zirconium deposits and imparts a distinct creamy or yellowish cast to the opacifier. What’s the real story? Titanium acts as a color modifier that interacts with iron particles, often amplifying the visual severity of existing impurities through lattice substitution.
How do trace elements interact?
The simultaneous presence of multiple transition metals creates complex color centers that are difficult to neutralize with standard masking agents. Consider this fact: understanding the specific chemical profile of your contaminants is the only way to effectively mitigate them.
| Element | Chemical Symbol | Visual Color Impact | |
|---|---|---|---|
| Iron Oxide | Fe2O3 | Red / Brown / Pink | |
| Titanium Dioxide | TiO2 | Cream / Yellow / Buff | |
| Chromium Oxide | Cr2O3 | Greenish / Grey tint |
Key Takeaway: Iron and Titanium are the primary chemical antagonists preventing pure whiteness in ceramics.
Sourcing Zirconium Silicate Raw Materials
Do geological origins dictate purity?
The specific geological location of the mineral deposit establishes the baseline purity potential, as sands from different regions carry distinct heavy metal fingerprints. It turns out that raw sands from specific coastal or inland deposits require vastly different levels of beneficiation to reach ceramic-grade standards.
- Mineralogical composition
- Extraction complexity
- Regional impurity profiles
Can natural variation be managed?
While natural geological variance is inevitable, it can be strictly controlled through rigorous batch selection and pre-processing blending strategies. Warning: sourcing from a single mine without third-party chemical verification invites dangerous volatility into your supply chain.
How risky are unverified sources?
Relying on unverified or spot-market sources often leads to receiving material with fluctuating mineralogy that disrupts automated production lines. You must realize that the initial cost savings of cheap raw sand are quickly erased by processing difficulties.
| Deposit Characteristic | Required Processing | Manufacturing Risk | |
|---|---|---|---|
| High Iron Content | Intense Magnetic Separation | High | |
| High Titanium Levels | Advanced Chemical Leaching | Moderate | |
| Clean Mineral Deposit | Standard Washing / Gravity | Low |
Key Takeaway: The geological source determines the processing complexity required to achieve a white product.
Optimizing Zirconium Silicate Washing Processes
Do chemical treatments improve hue?
Aggressive acid washing and chemical leaching are mandatory steps to strip surface-bound contaminants and significantly upgrade the sand’s intrinsic whiteness. Better yet, implementing multi-stage chemical washing can transform a standard industrial grade sand into a premium super-white ceramic grade.
- Hot acid leaching
- Gravity spiral separation
- Froth flotation
Is process consistency difficult?
maintaining identical chemical concentrations and washing durations across thousands of tons requires sophisticated automation and real-time monitoring. This is where it gets interesting: even a minor deviation in acid pH or wash time can result in a noticeable hue shift across an entire shipment.
Why is drying critical?
Proper drying techniques ensure that residual moisture does not lead to oxidation or agglomeration before the milling stage. Let’s look at the data: precise control over the washing and drying cycle is the primary defense against surface contamination.
| Process Step | Primary Function | Color Improvement Benefit | |
|---|---|---|---|
| Acid Wash | Dissolves surface oxides | Eliminates red/yellow tint | |
| Flotation | Separates mineral types | Removes grey impurities | |
| Thermal Drying | Removes moisture content | Prevents oxidation spotting |
Key Takeaway: Precision in chemical processing is just as critical as the initial quality of the raw material.
Milling Effects on Zirconium Silicate Opacity

Does particle size enhance whiteness?
Grinding zirconium silicate to a finer particle size drastically increases its refractive index and light scattering capabilities, resulting in superior whiteness and opacity. Ready for the good part? Optimized jet milling or ball milling can effectively mask minor chemical impurities by maximizing the surface area available for light reflection.
- Increased specific surface area
- Enhanced light scattering (Opacification)
- Improved glaze suspension
Can grinding media cause contamination?
Utilizing inferior grinding media, such as low-quality steel balls, introduces mechanical iron contamination that inevitably greys the final powder. Make no mistake: attempting to cut costs on grinding media will destroy the purity you achieved during the washing phase.
Is there an optimal grind size?
There is a specific “sweet spot” for particle size distribution where opacity is maximized without causing rheological issues in the glaze application. Ideally, you want a narrow particle size distribution that balances scattering efficiency with slurry stability.
| Variable | Impact on Whiteness | Potential Risk Factor | |
|---|---|---|---|
| Fine Grind (<5µm) | Increases (High scattering) | Agglomeration / Dusting | |
| Steel Media | Decreases (Grey tint) | Iron Contamination | |
| High-Alumina Media | Neutral / Positive | Higher Operational Cost |
Key Takeaway: Finer grinding improves whiteness through physics, but poor equipment introduces chemical contamination.
Thermal Impact on Zirconium Silicate Chemistry
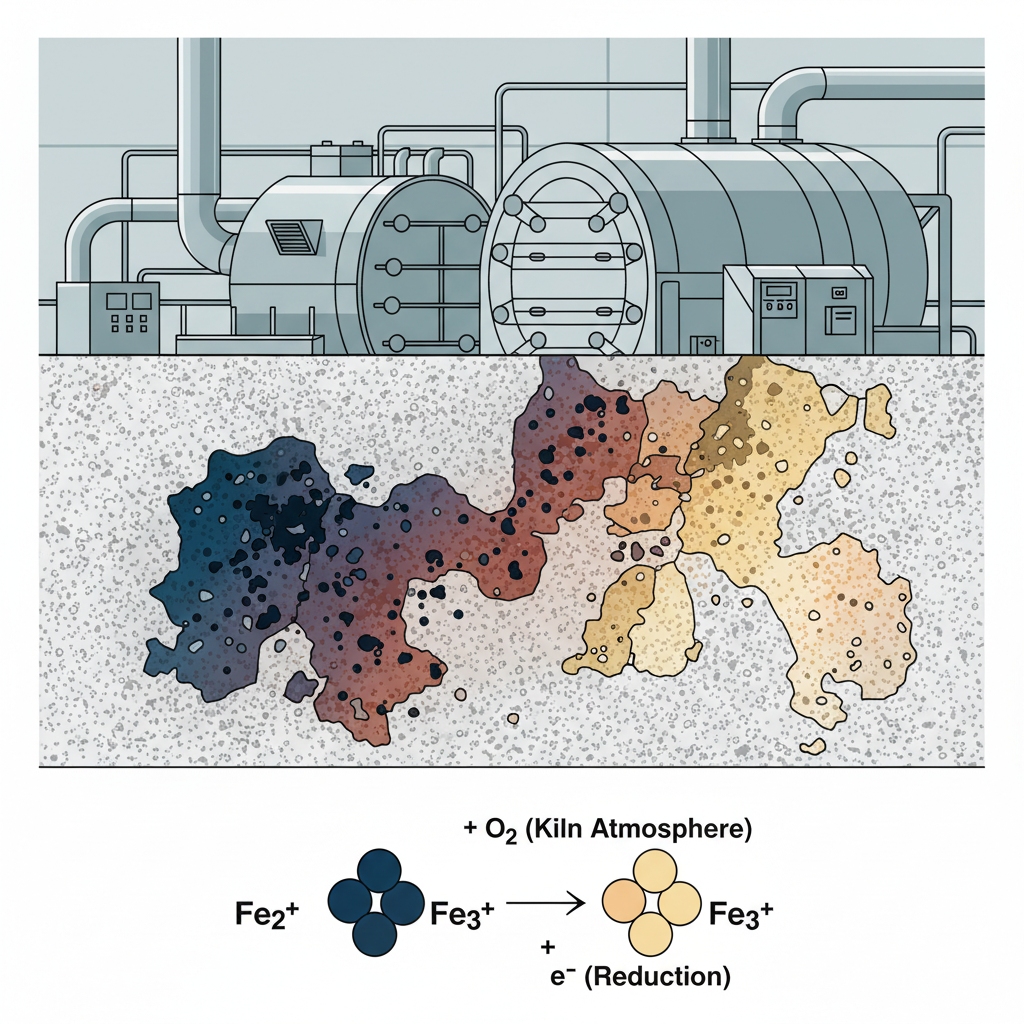
How does calcination stabilize color?
Calcination burns off residual organic matter and stabilizes the crystal lattice, but the kiln temperature profile must be meticulously calibrated to prevent defects. Here is the secret: the specific atmosphere inside the kiln controls the oxidation state of residual transition metals, locking in their final color.
- Organic contaminant removal
- Crystal phase stabilization
- Oxidation state control
What happens to iron at high heat?
Extreme heat forces iron impurities to transition between Ferrous (Fe2+) and Ferric (Fe3+) states, which manifest as drastically different colors in the final ceramic matrix. Briefly, maintaining a consistent oxidizing atmosphere ensures that iron remains in its least visible state.
Why is temperature control vital?
Fluctuating temperatures can lead to incomplete calcination or over-sintering, both of which degrade the optical properties of the zirconium silicate. The evidence shows that strict thermal management is the final gatekeeper of product quality.
| Temperature Zone | Iron Oxidation State | Visual Color Result | |
|---|---|---|---|
| <900°C | Incomplete Oxidation | Grey / Black spotting | |
| 900°C – 1200°C | Ferric (Fe3+) | Stable / Neutral White | |
| >1200°C | Matrix Interaction | Brownish fusion |
Key Takeaway: Thermal processing chemically alters impurities, permanently locking in the material’s color profile.
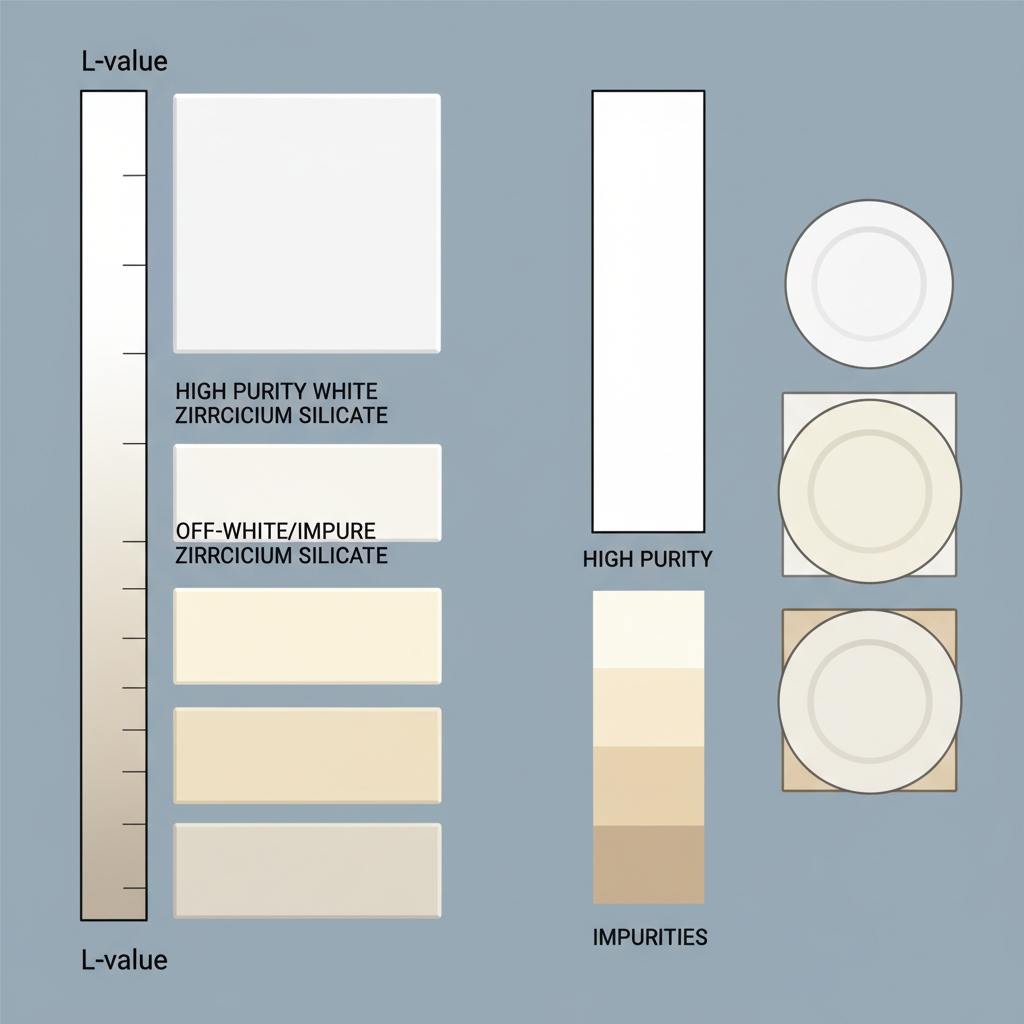
Measuring Zirconium Silicate Whiteness Specs

What is the CIE Lab standard?
The CIE Lab* color space is the global industrial standard for quantifying color through a three-dimensional coordinate system. To put it simply, it measures Lightness (L), Red/Green (a), and Blue/Yellow (b) to provide an objective, non-debatable numerical value for quality.
- L* (Lightness / Brightness)
- a* (Red to Green axis)
- b* (Yellow to Blue axis)
Why is the L-value the primary metric?
The ‘L-value’ specifically denotes brightness on a scale of 0 to 100, with higher numbers indicating a purer white closer to the theoretical ideal. You must understand that a drop in the L-value serves as an immediate early warning system for contamination, often before the human eye can detect a shift.
Can visual inspection be trusted?
Human visual inspection is subjective and prone to error due to lighting conditions and fatigue, making instrumental measurement indispensable. The reality is that reliance on “visual checks” alone is a recipe for quality control disputes.
| Metric | Technical Meaning | Ideal Target Range | |
|---|---|---|---|
| L* Value | Brightness (0-100) | > 95.0 | |
| a* Value | Red (+) / Green (-) | Near 0.0 | |
| b* Value | Yellow (+) / Blue (-) | Near 0.0 |
Key Takeaway: Objective data measurement is the only reliable method for assessing true material quality.
Zirconium Silicate Requirements for Ceramics

Do sanitary ware glazes need high purity?
Manufacturers of high-end sanitary ware and porcelain tiles rely on ultra-pure zirconium to act as a powerful opacifier that effectively hides the underlying clay body. Make no mistake: in the luxury sanitary market, even a slight grey cast is grounds for rejection, making purity the top procurement priority. For a deeper dive into the material’s application, read our article on the Application of Zirconium Silicate in Ceramics.
- Sanitary ware glazing
- Porcelain wall tiles
- Premium tableware
Is whiteness vital for industrial coatings?
For industrial coatings and paints, high whiteness ensures consistent opacity and reduces the number of coats required to achieve full coverage. You might not know that using consistent raw materials directly improves the chemical resistance and refractive efficiency of the final paint formulation.
How do different industries prioritize?
While all industries value consistency, the tolerance for variation differs significantly depending on the final application’s visual requirements. Consider this: a floor tile manufacturer might tolerate slight variances that a toilet manufacturer cannot.
| Industry Sector | Primary Requirement | Tolerance for Variation | |
|---|---|---|---|
| Sanitary Ware | Maximum Whiteness | Zero Tolerance | |
| Floor Tiles | Hardness & Durability | Low Tolerance | |
| Industrial Coatings | Opacity & Coverage | Moderate Tolerance |
Key Takeaway: High-end ceramics and coatings cannot function competitively without high-whiteness additives.
Analyzing Zirconium Silicate Cost Efficiency
Do production rejects inflate costs?
Using inconsistent raw materials leads directly to finished goods failing Quality Assurance, which exponentially increases waste disposal fees and energy consumption. Let’s be honest: purchasing cheaper, inconsistent zirconium silicate is a “false economy” that eventually drains your operational budget through rework.
- Wasted kiln energy
- Raw material loss
- Hazardous disposal fees
Is premium grade material worth the investment?
Investing in premium, chemically consistent grades eliminates the need for constant formulation adjustments, allowing you to streamline production and protect profit margins. Think about it: material stability allows you to fully automate your production lines and scale output with total confidence.
How does stability affect TCO?
The Total Cost of Ownership (TCO) for premium material is often lower than cheap alternatives when you factor in the elimination of downtime and scrap. The numbers show that paying a premium for purity pays for itself in yield improvements.
| Cost Factor | Cheap / Variable Material | Premium / Stable Material | |
|---|---|---|---|
| Upfront Material Cost | Low | High | |
| QA Rejection Rate | High (10-15%) | Low (<1%) | |
| Total Cost of Ownership | High | Low |
Key Takeaway: Consistent material prevents expensive production waste and unplanned downtime, securing long-term ROI.
Securing Reliable Zirconium Silicate Partners

What defines a top-tier supplier?
Leading suppliers utilize advanced purification methods like high-gradient magnetic separation and acid washing to physically remove color-altering impurities before bagging. To put it simply, these technologies scrub the material clean, ensuring the technical specifications on the datasheet actually match the powder you receive.
- Chemical acid leaching
- High-gradient magnetic separation
- Traceable supply chain
How should you choose a partner?
Select suppliers who provide transparent batch-specific Lab* data and can demonstrate total control over both their mining sources and purification lines. Here is a summary of the non-negotiable requirements you should demand in your next supply contract.
Why is transparency non-negotiable?
A supplier unwilling to share detailed quality data is likely hiding inconsistencies that will eventually become your production problem. Bottom line: transparency is the hallmark of a partner who is confident in their product’s performance.
| Critical Factor | Contract Requirement | Operational Benefit | |
|---|---|---|---|
| Purification Tech | Acid Wash + Magnetic | Removes Fe/Ti hazards | |
| Data Transparency | Lab* per batch | Guaranteed Specification | |
| Supply Chain | Traceable Mining Source | Long-term Stability |
Key Takeaway: Partner with suppliers like Global Industry who use advanced technology to guarantee purity.
Conclusion
Zirconium Silicate color variation is a multifaceted challenge driven by chemical impurities like iron and titanium, as well as critical processing variables such as milling particle size and calcination atmosphere. By understanding these root causes and prioritizing objective Lab* measurement over visual guesswork, you can eliminate production risks and ensure a flawless final product.
Contact Global Industry today to secure samples of high-whiteness Zirconium Silicate and bring unshakeable consistency back to your production line.
FAQ Section
Q1: Can I use Zirconium Silicate with a lower L-value for sanitary ware?
No, this is generally ill-advised; sanitary ware requires the highest purity (High L-value) because even minor grey or yellow tints are easily visible on curved white surfaces and will likely lead to product rejection.
Q2: Can I fix a batch that has been contaminated with iron during milling?
It is extremely difficult and costly; once iron from grinding media is mechanically introduced into the powder, it is hard to remove without re-processing the entire batch through magnetic separation, making prevention the only viable strategy.
Q3: Can I rely solely on visual inspection to check incoming raw materials?
No, visual inspection is too subjective and fails to detect subtle contamination; you should always require and verify Lab* data to ensure the material meets specific whiteness standards before it enters your production line.
Q4: Can I mix Zirconium Silicate from different suppliers to save money?
This is risky; different suppliers use different ore sources and processing methods, which can lead to unpredictable chemical interactions and inconsistent rheology in your glaze, causing more defects than the cost savings are worth.
Q5: Can I improve whiteness by just increasing the firing temperature?
Not necessarily; while heat affects color, exceeding the optimal temperature range (over 1200°C) can actually cause impurities to react adversely with the glaze matrix, potentially turning the finish brown or causing fusion defects.